SickleGenAfrica
About SickleGenAfrica
Vision
To align the survival of individuals who have sickle cell disease in Africa with national norms.
Mission
To develop prognostic biomarkers of organ damage through the discovery of key genetic modifying factors and cognate mechanisms, and build capacity and career pipelines in Africa to support patient-centred research to advance the development of innovative therapy for SCD.
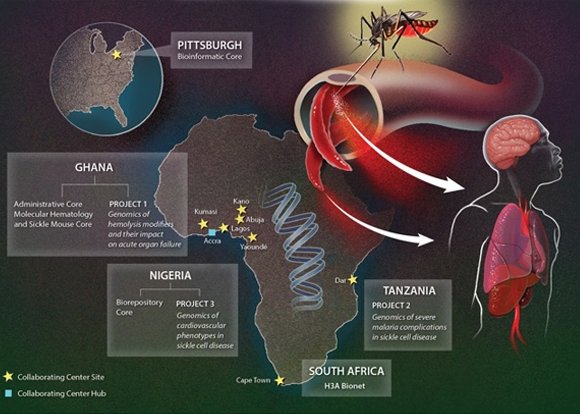
Nearly 2% of births in sub-Saharan Africa are affected with sickle cell disease (SCD). Bacteria-related deaths in SCD have reduced dramatically in children in the last 30 years in the West, and it is anticipated that a similar drop will occur in Africa once prevention and prompt management of infections becomes widely implemented on the continent.
During this same period, the death rate among adolescents and adults with SCD has not improved largely because we cannot predict, prevent and effectively manage the end-stage organ damage typical of this disease.
The SickleGenAfrica Network is made up of African scientists and international collaborators who are going to study 7,000 children and adults with SCD in Africa to identify genetic markers associated with the development of organ damage, with a special emphasis on the body’s defence against molecules released from damaged red blood cells that cause tissue injury. The long-term goal of the network is to develop strategies to predict, prevent and treat organ damage in SCD.
Leadership of SickleGenAfrica

Professor Solomon Ofori-Acquah
Director/ Principal Investigator
Professor Ofori-Acquah was appointed Dean of the School of Biomedical and Allied Health Sciences, University of Ghana in January 2017, and Associate Professor of Medicine and Human Genetics, University of Pittsburgh, USA in 2013. He is the Centre Leader of the West African Genetic Medicine Centre (WAGMC), and, Director and Principal Investigator of the Sickle Cell Disease Genomics Network of Africa (SickleGenAfrica).

Professor Gordon Awandare
Co-Principal Investigator
Gordon Awandare is an Associate Professor at the Department of Biochemistry, Cell and Molecular Biology of the University of Ghana. He is also the Director of the West African Centre for Cell Biology of Infectious Pathogens (WACCBIP). He obtained his BSc Biochemistry degree in 1998 and MPhil Biochemistry in 2002, both from the University of Ghana. He subsequently undertook his doctoral studies at the University Of Pittsburgh Graduate School Of Public Health, Pittsburgh, Pennsylvania, where he graduated with a PhD degree in Infectious Diseases and Microbiology in 2007, winning the Most Outstanding Student’s award in the process. Prof Awandare did his postdoctoral fellowship at the Malaria Vaccine Division of the Walter Reed Army Institute of Research in Silver Spring, Maryland from 2007-2010.

Professor Julie Makani
Co-Principal Investigator
Julie Makani is Associate Professor in the Department of Haematology and Blood Transfusion at Muhimbili University of Health and Allied Sciences (MUHAS) (http://www.muhas.ac.tz), the main clinical, academic and research centre in Tanzania. Tanzania has recognized sickle cell disease (SCD), as a major public health problem and it has been included as a priority condition in the national strategy for Non-Communicable Diseases in the Ministry of Health
Organizational Structure
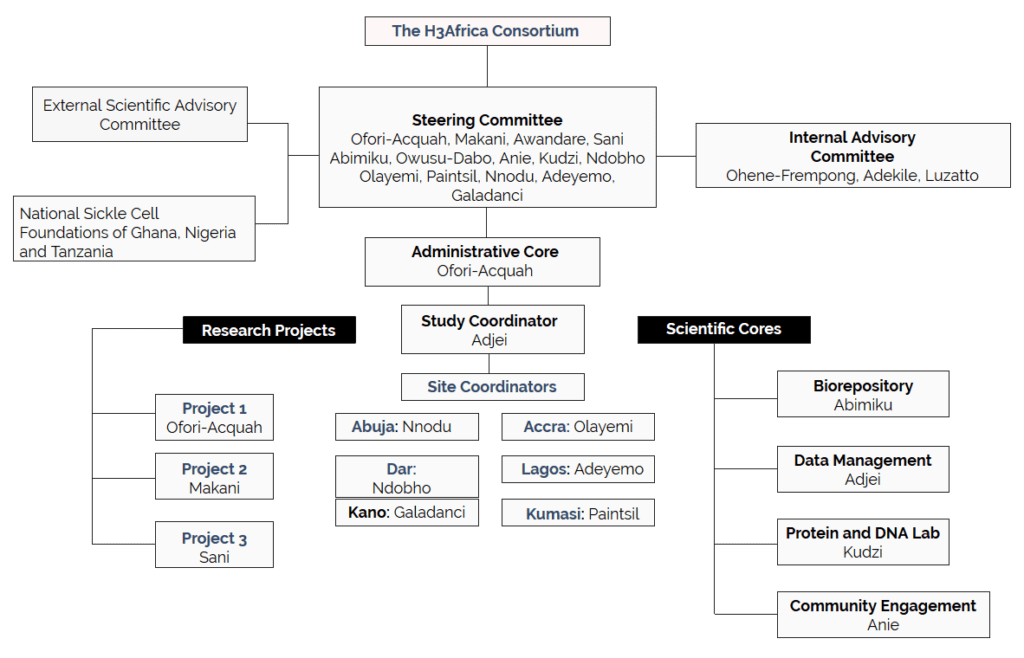
Site Coordinators

Professor Obiageli Nnodu
Site Coordinator - Abuja
Obi is the Site Coordinator for the Abuja Site and a Professor of Haematology and Blood Transfusion, Director, Centre for Sickle Cell Disease Research and Training University of Abuja (CESRTA), Honorary Consultant Haematologist, University of Abuja Teaching Hospital Gwagwalada, Abuja Nigeria. She is a founding member of Sickle CHARTA and Vice Chair of the Nigerian Sickle Cell Support Society (SCSSN) which works with the government and professional groups for the control of SCD in Nigeria. In that capacity, she is responsible for most of the in-country activities of the Network. She serves on technical committees of the government on non-communicable diseases and has completed a number of national assignments related to SCD. Obi is a member of the American Society of Hematology African Newborn Screening and Early Intervention Consortium while also a Chevening Scholar and alumnus of Imperial College London. She is Chair Advocacy of Sickle Pan African Network (SPAN).

Professor Edeghonghon Olayemi
Site Coordinator - Accra
Edeghonghon is the site coordinator for Accra. He is a graduate of the College of Medicine, University of Ibadan, Nigeria and a Consultant Haematologist. He is the immediate past Director of the Ghana Institute of Clinical Genetics (GICG), where the premier adult sickle cell disease clinic in Ghana is located. His areas of expertise include sickle cell disease, thrombosis and haemostasis. He is a Fellow of the Higher Education Academy, U.K. He coordinates all the activities at the Accra site and also represents the site at the site coordinators’ meeting.

Josephine Mgaya
Site Coordinator - Dar es Salaam
Josephine is the Site Coordinator at the Dar es Salaam site. She has ten years’ working experience in her field with an impressive appointment with the National Institutes of Health in the Laboratory of Early Sickle Mortality Prevention in the United States. She enjoys working hand in hand with other scientists to be able to bring about the answers in the management of SCD. Josephine has a BSc in Chemistry and Microbiology and a PgD in Haemoglobinopathy. She is currently working on studies involving intervention for individuals with Sickle Cell Disease such as early detection and management of complications, undergoing Hydroxyurea therapy. Her study will lead to an MSc by Research and Publication.

Dr. Najibah Galadanci
Site Coordinator - Kano
Najibah is an investigator and the site coordinator for the Kano, Nigeria site on the SickleGenAfrica: Sickle Cell Disease Genomics Network of Africa, where she oversees research activities, ensure protocol adherence, provide guidance for general clinic management of the study participants and also serve as a member of the steering and executive committee. She has been working in the department of hematology in Bayero University and Aminu Kano Teaching Hospital, northern Nigeria for over twelve years. Initially, as a resident doctor, she went on to become a lecturer and consultant hematologist. She is a practicing clinician and a laboratory physician in Kano, northern Nigeria. As a hematologist with a special interest in sickle cell disease, her research experience has been focused mainly on this area. She received the prestigious David Satcher Award, for a full tuition scholarship for a Masters in Public Health at Vanderbilt University (2013), and an NIH Forgarty fellowship award in June 2014. She concluded both the MPH program and the NIH Forgarty fellowship in May 2015. She was the site investigator of the first NIH funded Primary Prevention of Stroke in Children with Sickle Cell Disease in Nigeria trial referred to as the SPIN trial (1R21NS080639-01).

Dr. Vivian Paintsil
Director/ Principal Investigator
Vivian is the Coordinator for the Kumasi site. She oversees the organization of all project activities in Kumasi. Also, she is the clinical lead and sees to the daily running of the project. She completed KNUST with an MBChB and thereafter her fellowship training (FWACP), specialising in Paediatrics. She undertook a subspecialty training in Paediatric Haematology/Oncology in 2013 at the University of Cape Town; and has a certificate in Health Administration and Management. Vivian is a Specialist Paediatrician, and has been taking care of children with SCD for the past 14 years. She has been involved in the Newborn Screening Project at KATH and ORDISS, and is currently on the Sickle Pan African Research Consortium (SPARCo) project. It is her dream to have a foundation to take care of SCD affected children.

Dr. Titilope Adeyemo
Site Coordinator - Lagos
Titilope is the Site Coordinator for the Lagos Site and an Associate Professor in the Department of Haematology & Blood Transfusion, Faculty of Clinical Sciences, College of Medicine, University of Lagos and a Consultant Hematologist to the Lagos University Teaching Hospital, Nigeria. She earned her medical degree at the College of Medicine, University of Lagos, and completed her residency training in hematology at the Lagos University Teaching Hospital. She obtained a Master’s degree in Hemoglobinopathies at the University College London with a distinction. Her research interests are in sickle cell disease (SCD), blood transfusion science and inherited bleeding and clotting disorders. She is interested in the characterization of SCD based on genetic variations and impact on disease severity.
The H3Africa Consortium

Currently, there is an ongoing global effort to apply genomic science and associated technologies to further the understanding of health and disease in different populations, and, in particular, to identify individuals and populations who are at risk, due to genetic and/or environmental factors, for developing a specific disease. Possible outcomes of such research include early and more accurate diagnosis, the development of new drugs and potentially, personalized medicine - the systematic use of information about each individual patient to select or optimize the patient's preventative and therapeutic care.
However, most African countries are being left behind in this genomic revolution and if this is not urgently addressed, genomics will contribute to the widening of global and ethnic inequalities in health and economic well-being.
In response to this challenge, the African Society of Human Genetics, the National Institutes of Health (NIH) and the Wellcome Trust (WT) convened a Frontiers Meeting in Yaoundé, Cameroon in March 2009 to discuss a research agenda to study genetic diversity in health and disease in African populations. The meeting brought together scientific experts from a number of different fields to identify the major scientific questions, as well as the ethical and practical issues of pursuing a large-scale genomics research program in Africa. The key aim of the meeting was to engage with African scientists and other stakeholders in order to outline research priorities and discuss the appropriate study design needed to inform and ultimately impact health in the African populations. Following this meeting the concept of H3Africa was developed and the initiative was announced in June 2010. H3Africa is supported by the NIH Director's Common Fund as an initiative within its Global Health Program and by the Wellcome Trust as part of their continuing research portfolio in Africa.
For more information on the Consortium visit: www.h3africa.org

